Which of the Following Molecules Has the Highest Boiling Point
Of the diatomic elements H2 N2 O2 F2 Cl2 Br2 I2 all have dispersion forces. Toluene and phenol have similar boiling points.

Intermolecular Forces How Can I Determine The Highest Boiling Point Given A List Of Molecules Chemistry Stack Exchange
To predict boiling point we should have idea about van der waals forces which depend on surface area of the given mole View the full answer Transcribed image text.
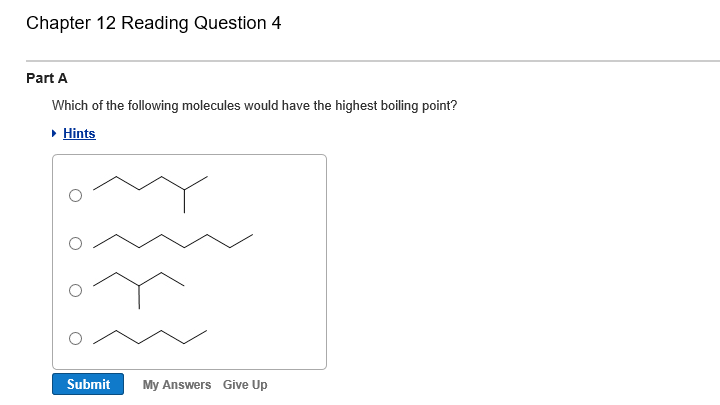
. QUESTION 10 Which of the following molecules has the HIGHEST boiling point. HCl will have a. CH4 is non-polar hence contain only weak van der waals forces hence it will have a low boiling point.
8 Does HF have a high boiling point. Cl straight chain molecule with the highest number of carbons has the highest boiling point. Hence CH3CH2CH2COOH has a greater boiling point than CH3CH2CH2OH.
2 1-Pentanol 138 C. 7 Why is the boiling point of HCl lowest while that of HF highest. HCl will have a lower boiling point than HF since F is more electronegative than Cl and possess a greater degree of hydrogen bonding.
CH3CH2CH344 amu 01 dipole moment CH3OCH346 amu 13 dipole moment CH3Cl50 amu 19 dipole moment CH3CHO44 amu 27 dipole moment CH3CN41 amu 39 dipole moment. 4 Which of the following has highest boiling point HF HCL. AnswerHF will have the highest boiling point.
Glucose usually melts at 146 C. As a result the magnitude of van der Waals forces of attraction decreases and hence the boiling decreases. 3 Painting 361 C.
CH3OH hydrogen bonding. 2 - Methyl pentane has the highest boiling point because the boiling point in alkane depends upon the chains of atoms and the molecular mass of the molecules. Which of the following organic bartleby.
Which of the following molecules has the highest boiling point AnswerHF will have the highest boiling pointExplanationCH4 is non-polar hence contain only weak van der waals forces hence it will have a low boiling point. Next comes methanol CH4O or CH3OH. Octane will have a higher boiling point than 2233tetramethylbutane because it branches less than 2233tetramethylbutane and therefore has a larger surface area and more van der Waals forces.
So the decreasing order of boiling point is C H 3 C H 2 C H 2 C H 2 C l C H 3 C H 2 C H 2 C l C H 3 C H C H 3 C H 2 C l C H 3 3 C C l. Propan-1-ol boils at a higher temperature than propan-2-ol. 1-Pentanol has the highest boiling point.
P is much less electronegative than both Cl and F hence PH3 has a much. CH4 London dispersion forces. View the full answer.
The other compounds in the options do not posses hydrogen bonds hence they will have much lower boiling points. Boiling points increase as the number of carbons is increased. Which of the following organic compounds has the highest boiling point.
4 Painting 103 C. 6 Which molecular substance will have the lowest vapor pressure. Which of the following has the highest boiling point.
Dimethyl ether CH3OCH3 is a polar molecule. Which of the following has the highest boiling point. These dimmers have a much higher boiling point than the corresponding alcohols due to stronger hydrogen bonds.
10 Why HCl has low. Alcohol are found to have boiling point approximately as 7837 C. It will have the next highest boiling point.
9 Why does HF have a higher boiling point than F2. What is the correct relation between acidic strength of primary secondary and tertiary alcohols. The more sphere like the molecule the lower its surface area will be and the fewer intermolecular Van der Waals interactions will operate.
Further because of two branches on the same carbon 22 dimethylbutane has lower surface area and hence lower. Butanoic acid OH O 22-dimethylbutane Pentanol Ethyl ethanoate. Sodium chloride has boiling point 1413 C.
Asked Sep 1 2019 in Chemistry by MangaLover general-chemistry. Which one of the following compounds will have the highest boiling point. A CH_3 CH_2 CH_3 CH_2 CH_5 B CH_3 CH_2 CH_3 CH_2 OH C CH_3 CH_2 CH_2 OCH_3 D CH_3 CH_2 CH_2 Cl E CH_3 CH_2 OCH_2 CH_3 Rank the following compounds in decreasing order to boiling points highest to lowest A IIIVIIII B IIVIIIII.
Branching decreases the boiling point. Now 2 methylpentane has the largest surface area and hence has the highest boiling point. This is because intermolecular forces often called van der Waals forces depend on attractions caused by quantum fluctuations in.
Nonane will have a higher boiling point than octane because it has a longer carbon chain than octane. So it is easy to identify that NaCl sodium chloride has highest boiling point amongst the given choices. AmmoniaNH3 has the highest boiling point at -3334 C while Methane CH4 has a boiling point of -1616 C and Hydrogen sulfide SH2 has a boiling point of -60 C.
The boiling point of butane is close to 0 degrees Celsius whereas the higher boiling point of butanone 796 degrees Celsius can be explained by the shape of the molecule which creates an attractive force between the oxygen on one molecule and the hydrogen on. Of the two molecules shown in the figure one would expect that CO2 has a higher boiling point than NO2 because CO2 is linear and nonpolar. Straight-chain molecules have higher boiling point than the branched-chain compounds.
For isomeric alkyl halides boiling point decreases as branching increases. 5 Which has higher boiling point HF or HCL. Water boils at 100C.
Pentanol has a higher boiling point compared to others because it contains stronger intermolecular interactions known as. CH3OCH3 dipole-dipole attractions. It has the highest boiling points.
This is because Ammonia can form strong hydrogen bonds but which makes it. THIS IS THE BEST ANSWER. As the branching increases the surface area decreases.
The boiling points of the given compounds are as follows 1 2-Pentanone 101 C. Therefore the element with the greatest total number of electrons will have the highest boiling point iodine and the element with the smallest total number of electrons will have the lowest boiling point hydrogen. Methanol has strong hydrogen bonds.
Stereoisomers formed by ring formation at the carbon which was originally a carbonyl aldehyde or ketone in the open chain form of monosaccharides. CaCO3 is an ionic compound.
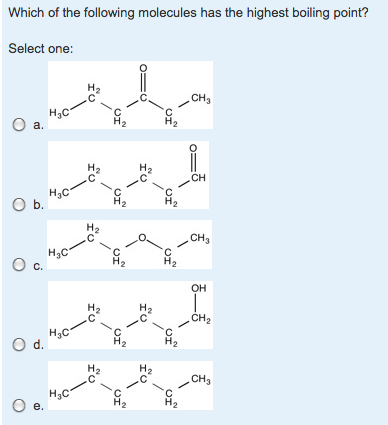
Solved Which Of The Following Molecules Has The Highest Chegg Com

Answered 16 Which Of The Following Molecules Bartleby

Solved Which Of The Following Molecules Would Have The Chegg Com
Comments
Post a Comment